|
2/17/2024 0 Comments Reactivity trend in alkaline earth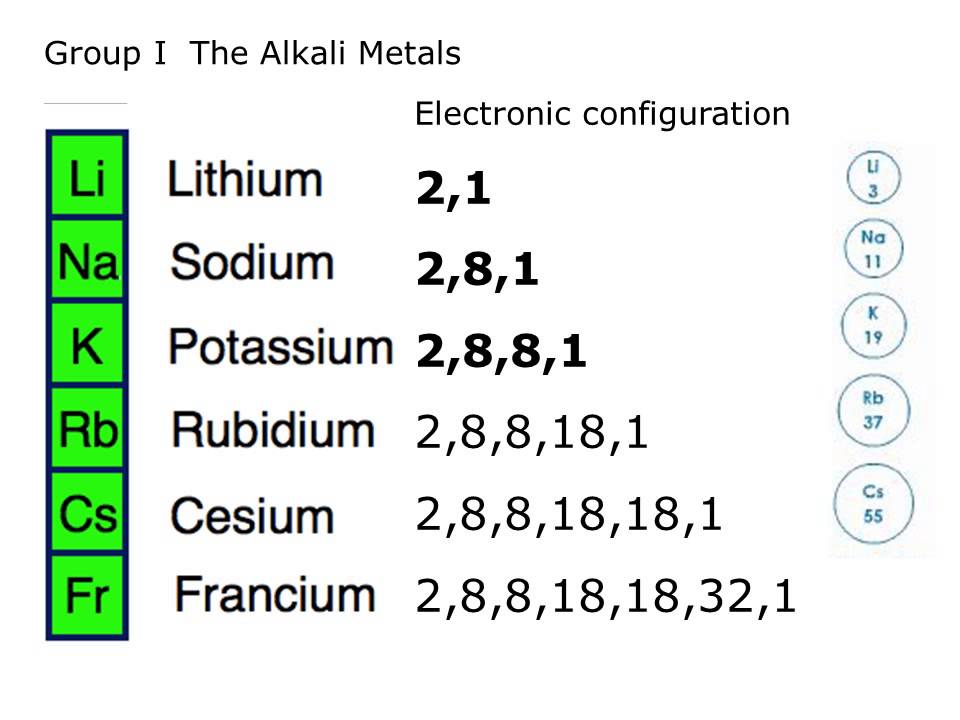
They react easily with most non-metals, including the halogens. Lithium gets a layer of lithium nitride when exposed to air. When exposed to air, these metals, particularly potassium, can form unstable peroxides and superoxides over time, which can explode in the right circumstances. The reaction of these metals with water produce hydrogen and the corresponding hydroxide in solution. Alkali Metal ChemistryĪlkali metals form compounds almost exclusively in the +1 oxidation state. The alkali metals include lithium, sodium, potassium, rubidium, cesium, and francium. It has the lowest electronegativity and the lowest first ionization energy. Of the 5 non-radioactive alkali metals, cesium has the lowest melting and boiling point, the highest density, the largest atomic radius, and the highest reactivity. Rubidium has a striking red-violet flame color. They also have low electronegativities, low first ionization energies, but higher second ionization energies.īecause the outer s electron is easily excited, these metals tend to have very distinctive flame colors. However, recent research shows that it is in fact not hydrogen, but a Coulomb explosion responsible for the explosive behavior in water.Īll of them have a very low density, and in fact lithium, sodium, and potassium float on water. Most textbooks explain the violent reaction as hydrogen catching on fire. The heavier elements react more violently with water, but even sodium can cause a large explosion if sufficient quantity is used. They are usually stored under argon, or in mineral oil. 
They react quickly with air, and violently with water. Because of their weak metallic bonding, they have lower melting points than other metal groups. The group 1 elements are all shiny, soft, and highly reactive. In 1807, Sir Humphry Davy, soon after Volta first invented a battery, isolated sodium and potassium metal by electrolysis of molten sodium hydroxide and potassium hydroxide. These metals have very similar properties and group trends.Īlkali metals have been known for a long time. They all have one electron in their outer shell, which results in weak metallic bonding. 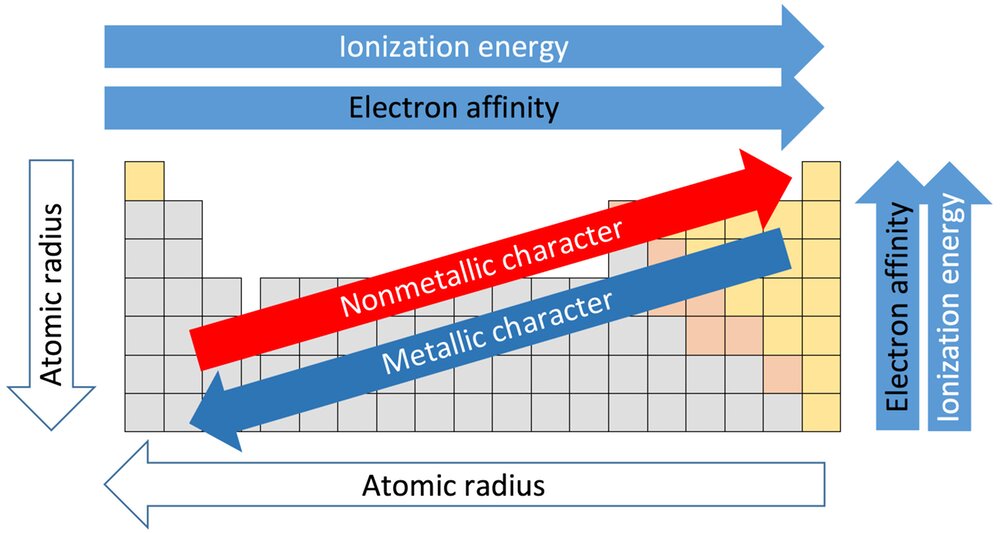
They include lithium, sodium, potassium, etc. This is the first column of the periodic table. The alkali metals are all members of group 1 on the periodic table, minus hydrogen. In chemistry, it refers to a base that can dissolve in water. The word “alkali” comes from Arabic, meaning “ashes of the saltwort”.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
